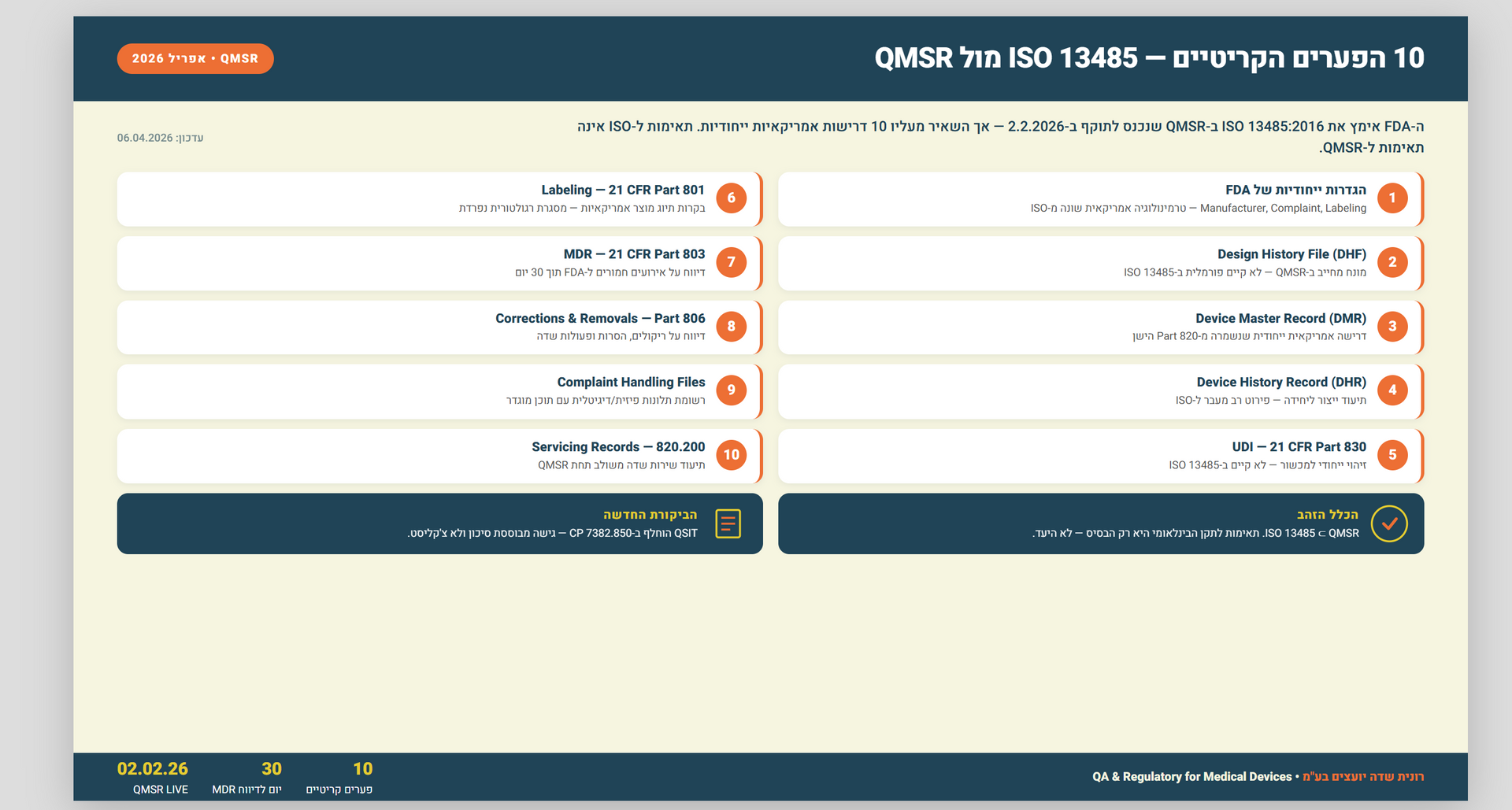
Understanding the Differences in Processes between External Audits and FDA Inspections
Introduction:
External audits and FDA inspections are vital processes that ensure compliance and quality control in regulated industries like healthcare and pharmaceuticals. While both serve the purpose of evaluating an organization's practices and adherence to standards, they differ significantly in their approach, scope, and objectives. This article aims to provide comprehensive information on the key differences in processes between external audits and FDA inspections for a better understanding.
Section 1: Purpose and Focus
External Audits:
External audits are conducted by independent third-party auditing firms or professionals hired by organizations. Their primary objective is to assess financial records, operations, and internal control systems. These audits focus on financial accuracy, risk assessment, regulatory compliance, and operational efficiency. External audits help organizations ensure the integrity and transparency of their financial statements, identify areas of improvement, and provide assurance to stakeholders such as investors, creditors, and shareholders.
FDA Inspections:
FDA inspections, conducted by the U.S. Food and Drug Administration (FDA), ensure compliance with regulatory requirements in industries such as pharmaceuticals, medical devices, biologics, and food. These inspections primarily evaluate product safety, efficacy, manufacturing processes, quality control systems, labeling, and adherence to Good Manufacturing Practices (GMP) guidelines. FDA inspections aim to protect public health by ensuring that regulated products meet the required standards of safety, effectiveness, and quality.
Section 2: Regulatory Authority
External Audits:
External audits are not mandated by any specific regulatory authority. They are conducted based on industry best practices, contractual requirements, or to meet stakeholder expectations. The scope and frequency of external audits are determined by the organization or relevant stakeholders. External audits may be conducted to comply with legal or regulatory requirements, to meet the needs of investors or lenders, or as part of an organization's risk management and internal control processes.
FDA Inspections:
FDA inspections are carried out by the FDA, a federal agency responsible for protecting public health by ensuring the safety and effectiveness of regulated products. The FDA has the authority to inspect regulated facilities, collect samples, review documentation, and take enforcement actions for violations or non-compliance. FDA inspections are mandated by regulatory frameworks such as the Food, Drug, and Cosmetic Act (FD&C Act) and are essential for maintaining the safety and integrity of the regulated industries.
Section 3: Frequency and Notification
External Audits:
The frequency of external audits varies based on organizational policies, contractual obligations, or industry standards. Organizations may choose to conduct external audits annually, biennially, or as needed. The timing and scope of external audits are typically known in advance, allowing organizations to prepare and provide requested documentation. External audits may also be triggered by significant events such as mergers or acquisitions, changes in leadership, or financial irregularities.
FDA Inspections:
FDA inspections are unannounced and can occur at any time. They may be routine or triggered by events like product recalls, complaints, adverse events, or emerging safety concerns. The element of surprise helps assess an organization's day-to-day compliance without prior preparation. FDA inspections are conducted with the aim of evaluating the ongoing compliance of regulated facilities and ensuring that the products meet the required standards throughout their lifecycle.
Section 4: Scope and Documentation
External Audits:
External audits primarily focus on financial records, internal controls, and compliance with accounting standards. Auditors review financial statements, transaction records, policies, procedures, and related documentation. The audit process may involve interviews with key personnel, sample testing, and evaluation of risk management practices. External audits provide an independent assessment of an organization's financial health, accuracy of financial reporting, and effectiveness of internal controls.
FDA Inspections:
FDA inspections have a broader scope and concentrate on product quality, safety, and compliance with regulations. Inspectors review facility design
Inspectors review facility design, equipment qualification, validation of manufacturing processes, storage and handling practices, recordkeeping, adverse event reporting, and product labeling. They may request access to batch records, quality control data, distribution records, complaint files, and other relevant documentation. FDA inspections aim to ensure that regulated facilities adhere to the applicable regulations and guidelines, maintain proper documentation and records, and have robust quality systems in place to safeguard the public from potential risks associated with regulated products.
Section 5: Process Steps
External Audits:
The process of external audits typically involves several steps:
- Planning: The audit is planned in advance, considering the scope, objectives, and timeline. The audit team identifies the key areas to be assessed and determines the necessary resources.
- Fieldwork: Auditors conduct on-site visits to the organization being audited. They gather evidence, review documents, and perform interviews with relevant personnel. Sample testing may be conducted to evaluate the effectiveness of internal controls and identify potential risks.
- Analysis and Reporting: The collected information is analyzed, and findings are documented in an audit report. The report outlines any deficiencies, non-compliance, or areas requiring improvement. Recommendations may be provided to address identified issues.
- Follow-up: Organizations are expected to take corrective actions based on the audit findings. External auditors may conduct follow-up audits to ensure the implementation of recommended changes.
FDA Inspections:
FDA inspections follow a structured process defined by the FDA. The key steps in an FDA inspection include:
- Notice of Inspection: Regulated facilities receive an unannounced visit from FDA inspectors, who present their credentials and provide a Notice of Inspection (Form FDA 482). This notice informs the facility of the purpose and scope of the inspection.
- Inspection Activities: Inspectors conduct a thorough evaluation of the facility, processes, and records. They review documentation, conduct interviews, collect samples, and observe operations. Inspectors may also perform on-site testing or verification activities.
- Observations: During the inspection, FDA inspectors document observations of any deficiencies, violations, or non-compliance with regulations. These observations are often communicated to the facility's management during the inspection or in a subsequent Establishment Inspection Report (EIR).
- Response and Corrective Actions: Following the inspection, the facility has the opportunity to respond to the observations and provide corrective actions to address identified issues. The FDA may review and assess the adequacy of the facility's response and the effectiveness of the proposed corrective actions.
- Follow-up and Compliance: In cases where significant violations or non-compliance are found, the FDA may initiate follow-up inspections or take enforcement actions, such as issuing warning letters, imposing fines, or initiating legal proceedings.
Section 6: Impact on Organizations
External Audits:
External audits provide organizations with valuable insights into their financial processes, internal controls, and compliance with regulations. They help organizations identify areas for improvement, enhance transparency and accountability, and build trust with stakeholders. Successful external audits can positively impact an organization's reputation, investor confidence, and access to capital.
FDA Inspections:
FDA inspections have a significant impact on regulated industries. Compliance with FDA regulations is essential for market access, product approval, and ongoing operations. Successful FDA inspections validate an organization's commitment to product quality and safety, enhance its reputation, and ensure continued compliance with regulatory requirements. Non-compliance with FDA regulations can lead to product recalls, regulatory actions, damage to reputation, and potential financial and legal consequences.
Conclusion:
External audits and FDA inspections serve distinct purposes in evaluating compliance and quality control in regulated industries. External audits focus on financial accuracy, risk assessment, and operational efficiency, while FDA inspections primarily ensure product safety, efficacy, and compliance with regulations.
Here are some authoritative sources that provide in-depth information about the difference between external audits and FDA inspections:
- U.S. Food and Drug Administration (FDA):
- Official website: www.fda.gov
- The FDA's website offers comprehensive information about FDA inspections, regulations, compliance, and industry-specific guidance.
- Public Company Accounting Oversight Board (PCAOB):
- Official website: www.pcaobus.org
- The PCAOB provides oversight and sets auditing standards for external audits of public companies. Their website offers resources, publications, and guidance related to external audits.
- International Organization for Standardization (ISO):
- Official website: www.iso.org
- ISO develops and publishes international standards, including those related to auditing and quality management systems. Their website provides access to standards, guidelines, and publications related to external audits.